With the recent FDA delay, what's the deal with Sarepta Therapeutics. Seems like a sure thing but then why would the FDA delay their decision and request more data.
A good look at their clinical trail is in the WSJ this week. Where's the drug FDA?
The Food and Drug Administration is sitting on a therapy for Duchenne muscular dystrophy, and the agency may have days to waste but the boys don’t. Bureaucratic malpractice on a safe and effective treatment is corroding the agency’s scientific credibility and the public’s trust.
FDA in May delayed a decision on eteplirsen by Boston-based Sarepta Therapeutics. There is no treatment for Duchenne, a fatal disease that claims a boy’s ability to walk before organ failure in his 20s. Eteplirsen jumps over genetic code to produce a missing protein known as dystrophin.
Read more at WSJ(FYI, if you google the link title you can get around their purchase options sometimes)
A look at their stock profile: $SRPT
I have enjoyed CNBC Meg Terrill's Look at Biopharma lately as well with the upcoming elections and terrible year that Biotech has been having.
Check out more videos of Meg here. http://www.cnbc.com/meg-tirrell/
Stock investments ranging from Biotech, Pharmaceutical, and Medical Devices in the Healthcare Sector. Covering Clinical Trial recommendations and FDA Approvals.
Showing posts with label FDA. Show all posts
Showing posts with label FDA. Show all posts
7/6/16
Sarepta Therapeutics: What Now After FDA Delay?
Labels:
approval,
Biotech,
catalysts,
Clinical trial,
cnbc,
FDA,
meg terrill,
muscular dystrophy,
Pharmaceuticals,
sarepta,
sarepta therapeutics,
srpt,
Stock,
stock quote
11/16/10
Human Genome Sciences Gains FDA Recommendation for Benlysta. Shares Soar 10% in Afterhours Trading
Good news today for Human Genome Sciences and those who suffer from Lupus. FDA voted in favor of Benlysta with a strong vote of 13-2. Although the FDA doesn't always approve drugs that are recommended by the panel, many times they do, and more so with an Approval Recommendation than without one. Shares of Human Genome Sciences were up 10.51% to 28.60 in afterhours trading.
Trading was halted throughout the day and will resume normal trading tomorrow. I expect a strong day tomorrow and it should be watched closely tomorrow. It's partner Glaxo SmithKline was trading slightly higher at 39.45 +0.91 a 2.36% gain. This is really good news for investors who have watched HGSI drop from a high of 30.18 in September to a low of 23.60 this past Friday from a Doctor scandal, a CRL for Zalbin, and safety concerns from their Phase III clinical trial results for Benlysta.
It did report positive 3Q 2010 results in the following press release, Human Genome Sciences Announces Third Quarter 2010 Financial Results and Key Developments. I see HGSI stock to rise towards the 32-35 mark with possible Full Approval coming December 9th.
Please follow BioPharma Investor for more Biotech and Pharmaceuticals News and FDA recommendations. I do my best to provide up to date news and extensive stock research in the BioPharma field. Subscribe in a Reader powered by Feedburner or join to have updates sent to your email. It's all free.
Yahoo Finance--HGSIGoogle Finance--HGSI
Yahoo Finance--GSK
Google Finance--GSK
FDA panel favors lupus drug from Human Genome Sciences despite mixed results in studies
Labels:
Belimumab,
Benlysta,
FDA,
FDA Recommendation,
Glaxo SmithKline,
GSK,
HGSI,
Human Genome Sciences,
Lupus,
Lupus Foundation of America
10/24/10
Jim Cramer Makes Mistakes Too
 Image by Michael McDonough via FlickrJim Cramer apologizes for his mistakes. Jim Cramer isn't always right on his calls so just take that in mind when you watch Mad Money. But the guy is entertaining And sometimes he is right and sometimes he is wrong. I too didn't see this decision by the FDA happening and apologize myself but I only liked Alkermes as a trade and still think it is a buy right now. Alkermes symbol ALKS. Eli Lilly (LLY) also posted strong 3Q earnings and was up Friday.
Image by Michael McDonough via FlickrJim Cramer apologizes for his mistakes. Jim Cramer isn't always right on his calls so just take that in mind when you watch Mad Money. But the guy is entertaining And sometimes he is right and sometimes he is wrong. I too didn't see this decision by the FDA happening and apologize myself but I only liked Alkermes as a trade and still think it is a buy right now. Alkermes symbol ALKS. Eli Lilly (LLY) also posted strong 3Q earnings and was up Friday. 4 Rules for Trading Biotech Spec Stocks
Two stocks that Cramer recommended last Thursday, Oct. 14 got crushed this week after an expected Food & Drug Administration approval never happened.
Everyone, including the “Mad Money” host, assumed regulators would bless a new diabetes medication being made by Amylin [AMLN 11.30 -0.18 (-1.57%) ] and Alkermes [ALKS 11.07 -0.03 (-0.27%) ]. But when the FDA on Tuesday requested more data rather than approving the drug, their share prices plummeted 50 percent and 30 percent, respectively.
So where did Cramer, and even the most negative of analysts, go wrong? What lessons has he learned from this week? He broke them down into four takeaways for any investors considering a little biotech speculation going forward. Watch the video 4 Rules For Trading Biotech for his full report.
Related articles
- 4 Rules for Trading Biotech Spec Stocks - CNBC (news.google.com)
- F.D.A. Rejects New Diabetes Drug (prescriptions.blogs.nytimes.com)
- Amylin, Alkermes Once-Weekly Diabetes Drug Fails to Win FDA Approval (xconomy.com)

Labels:
Alkermes,
Amylin Pharmaceuticals,
Eli Lilly,
FDA,
Food Drug Administration,
Jim Cramer,
Mad Money
10/23/10
Put Calls Anyone?
 Image via Wikipedia Arena Pharmaceuticals is in a tough spot as the FDA rejects their weight loss drug lorcaserin, apparently one of the safer of the 3 up for approval.
Image via Wikipedia Arena Pharmaceuticals is in a tough spot as the FDA rejects their weight loss drug lorcaserin, apparently one of the safer of the 3 up for approval.Here is the link to investors.
This decision was expected and predicted in my past articles on the Obesity Drug Market. We all saw it coming. I'm sorry if you didn't. But expect loses for Orexigen, and Vivus with their upcoming FDA decisions. I can only see gains made with shorting the stocks. Those that had are sitting pretty right now. It's a gamble but sometimes these gambles pay off. I expect more loses to mount for these companies in the near future. Arena Pharmaceuticals, Vivus Inc., and Orexigen Therapeutics.
What they chose to do next is anyones guess. The technology could be sold off for further research. And the company enters bankruptcy court. Nonetheless just passing on the news. I'm not always right but some are easier to pick than others. Last weeks news of Amylin, Alkermes, and Eli Lilly and Company for their diabetes medicine was a heartbreaker for those who thought it would go the other way. But there are always more to look at. Keep looking forward and use it as a lesson.
Arena Pharmaceuticals Enters FDA Purgatory
Welcome to FDA purgatory, Arena Pharmaceuticals. It could be a long wait.
Labels:
Anti-obesity medication,
Arena Pharmaceuticals,
ARNA,
Clinical trial,
FDA,
FDA Decisions,
Food and Drug Administration,
Lorcaserin,
Orexigen Therapeutics,
Vivus
10/20/10
The Short-Seller Who Got Amylin Right
 Image via WikipediaNEW YORK (TheStreet) -- Not all investors are shocked and upset about the decision by U.S. regulators to reject Amylin Pharmaceuticals' (AMLN) diabetes drug Bydureon. Short-sellers -- investors who bet that Amylin shares would fall -- are positively giddy and making money.
Image via WikipediaNEW YORK (TheStreet) -- Not all investors are shocked and upset about the decision by U.S. regulators to reject Amylin Pharmaceuticals' (AMLN) diabetes drug Bydureon. Short-sellers -- investors who bet that Amylin shares would fall -- are positively giddy and making money. Amylin shares are down $10.35, or 50%, to $10.14 in Wednesday trading.
How did the shorts get Amylin right when a majority of investors believed Bydureon was going to be approved? I spoke with one Amylin short early Wednesday about his bear thesis. This hedge fund manager asked to remain anonymous because the rules of his fund forbid him to speak publicly about his investments.
"I always believed that Amylin and Eli Lilly (LLY) were taking shortcuts to get Bydureon through the FDA" and the agency was going to have problems with this approach, said this short seller.
Labels:
Alkermes,
Amylin Pharmaceuticals,
Eli Lilly,
FDA,
FDA CRL,
Food and Drug Administration,
New Drug Application,
TheStreet
10/14/10
Blockbuster FDA Decisions In The Next Few Months--FDA Calendar Predictions
FDA's Upcoming Blockbuster Drug Decisions for Q4 2010
October 11, 2010
Again I am predicting the upcoming FDA Calendar for the next upcoming months for the Fourth Quarter. This is just an initial walkthrough of all the upcoming decisions. Lets see how well I do. I am doing pretty well from October's FDA calendar.
The FDA has some of its biggest decisions of the year looming in the next few months. The first new lupus drug in half a century, a blood-thinner with mega-blockbuster potential, controversial weight drugs and more are all up for regulatory decisions that can move markets and either throw a bucket of cold water or high octane gasoline on stock prices.
In every case, BioPharma Investor has been following the deals and data every step of the way--from early-stage development right through to the NDA and deal-making phases. Now everyone faces the moment of truth, or at least a complete response letter.
The FDA's final decision may be signaled by an expert panel vote, but this is one area where there really is no sure thing. The agency has appeared to be taking a tough stance on safety, which has bedeviled some late-stage therapies. And its complete response letters are always opened with a mix of dread and hope. We've seen plenty of surprising decisions so far this year, and there will likely be more in the months ahead.
I have to do some more research on my Approval Decisions---Here is an preliminary list of FDA Approvals coming in the 4th Quarter of 2010. This is just an estimated guess off the top of my head. Some are toss-ups. The FDA is leaning more on the Complete Response Letter side which isn't necesarily a death sentence as why the drug was not approved and how it might be possible to get approval. Usually, it requires more Clinical Trials when a company receives a CRL from the FDA.
If you would like more Subscribe To BioPharma Investor for everyday stock news and up to date FDA decisions. Biopharma Investor
FDA Calendar Predictions
October 11, 2010
Again I am predicting the upcoming FDA Calendar for the next upcoming months for the Fourth Quarter. This is just an initial walkthrough of all the upcoming decisions. Lets see how well I do. I am doing pretty well from October's FDA calendar.
The FDA has some of its biggest decisions of the year looming in the next few months. The first new lupus drug in half a century, a blood-thinner with mega-blockbuster potential, controversial weight drugs and more are all up for regulatory decisions that can move markets and either throw a bucket of cold water or high octane gasoline on stock prices.
In every case, BioPharma Investor has been following the deals and data every step of the way--from early-stage development right through to the NDA and deal-making phases. Now everyone faces the moment of truth, or at least a complete response letter.
The FDA's final decision may be signaled by an expert panel vote, but this is one area where there really is no sure thing. The agency has appeared to be taking a tough stance on safety, which has bedeviled some late-stage therapies. And its complete response letters are always opened with a mix of dread and hope. We've seen plenty of surprising decisions so far this year, and there will likely be more in the months ahead.
I have to do some more research on my Approval Decisions---Here is an preliminary list of FDA Approvals coming in the 4th Quarter of 2010. This is just an estimated guess off the top of my head. Some are toss-ups. The FDA is leaning more on the Complete Response Letter side which isn't necesarily a death sentence as why the drug was not approved and how it might be possible to get approval. Usually, it requires more Clinical Trials when a company receives a CRL from the FDA.
If you would like more Subscribe To BioPharma Investor for everyday stock news and up to date FDA decisions. Biopharma Investor
FDA Calendar Predictions
Labels:
2010 Drug Approvals,
Amylin,
AstraZeneca,
AZN,
Biotech,
BMY,
Eli Lilly and Company,
FDA,
FDA Calendar,
HGSI,
Human Genome Sciences,
Pharmaceuticals,
Q4 Drug Approvals
10/12/10
Big Gains For Pain Therapy Stocks Today: KG, PTIE, WPI, ENDP
So Pfizer is increasing their portfolio with King Pharmaceuticals, a maker of veterinary meds and pain therapies. In addition to this news, another company came into the spotlight for pain management therapies. This company is Pain Therapeutics (PTIE). Pain Therapeutics is in clinical trials now in collaboration with King Pharmaceuticals so look for Pfizer to kick in some clinical trial expertise with Pain Therapeutics (PTIE). Additional companies involved in pain therapies is Endo Pharmaceuticals. I highly suggest buying Endo as a buy right now. Since June 2010 Endo's stock has risen form 20.22 to its current trading at 34.99. I suggest a limit order to buy at some discount. Another Pain therapies stock is Watson Pharmaceuticals, a generic drug maker.
My opinion on this is that Pain Therapeutics could be slightly overvalued on the hyped news, and although it may seem attractive to buy right now, I suggest waiting for the pullback as it will be years before these clinical trials are actually approved by the FDA. Also, I am a little cautious placing a buy on Pfizer right now as they will be losing patents on Lipitor next year and other brand name drugs will become available as generics. I might actually place Pfizer's stock right now on hold or sell, as earnings could be lower than expected. Just because everyone is doing it does not make it a buy in my opinion. I actually do not necessarily see King Pharmaceuticals as such a hot buy with only 4.85% profit margin and 18M in profits on 380M in revenue. But given that layoff will presume who knows.
Stick to your guns and remain a cautious investor and good things will come. I apply Warren Buffett's principles to the BioPharma Stock World and like purchasing stocks at a discount. You should be searching for the next Pfizer and not chasing after something that is overly hyped. Just my opinion.
Stocks mentioned in this article
Pfizer--(PFE)
King Pharmaceuticals --(KG)
Endo Pharmaceuticals --(ENDP)
Pain Therapeutics --(PTIE) Up 18.56% today on the news
Watson Pharmaceuticals--(WPI)
Labels:
Alexza Pharmaceuticals,
Biotechnology and Pharmaceuticals,
Endo Pharmaceuticals,
FDA,
Geron,
King Pharmaceuticals,
Pain management,
Pain Therapies,
Pfizer,
TheStreet,
Warren Buffett
10/11/10
Disappointing Day For Alexza Pharmaceuticals
You really have to know what you are doing though when you make projections like this as is the drugs are approved then you will not be too happy with the results. It's really a difficult line to cross but I see the value of Shorting Stocks under the Obama FDA panel. The panel has rejected 4 drugs so far in October.
That doesn't mean that you should have shorted Hospira or Human Genome Sciences (HGSI) because these are large and mid cap stocks that are affected less by these FDA decisions. Human Genome has an upcoming drug approval in December for their Lupus therapy and I actually like the stock right now and consider it a buy. Alexza and Jazz just had too many red flags for me for approvals. You have to do your homework and look through the clinical trial data available. Or just follow BioPharma Investor and let me give you helpful advice on these decision.
Wikinvest--Alexza Pharmaceuticals (ALXA) Closed: 1.35 Down 1.65 for a 54.46% Loss
Wikinvest--Jazz Pharmaceuticals (JAZZ) Jazz will probably will be going down with today's CRL.
Jazz Pharmaceuticals Receives FDA Complete Response Letter Regarding JZP-6 for Treatment of Fibromyalgia
FDA O-Fer October Drug Approvals
BOSTON (TheStreet) -- U.S. regulators have to approve at least one new drug in October, right?
The U.S. Food and Drug Administration has a perfect 0-4 record so far this month -- four drug approval decisions, four drug rejections. Monday, Alexza Pharmaceuticals(ALXA) and Jazz Pharmaceuticals(JAZZ) received the bad news. Last week, it was FDA rejections for Human Genome Sciences(HGSI) and Hospira(HSP).
Perhaps the best shot for more positive news will come Tuesday when Alkermes(ALKS) is expecting FDA to rule on its drug for opioid addiction.
Labels:
Alexza Pharmaceuticals,
ALXA,
AZ-004,
Clinical trial,
CRL,
FDA,
Food and Drug Administration,
GERN,
Geron,
Jazz,
Jazz Pharmaceuticals,
TheStreet,
Valeant Pharmaceutical,
VRX
8/20/10
Jazz Pharma rejected for Fibramyalgia Drug by FDA vote 20-2
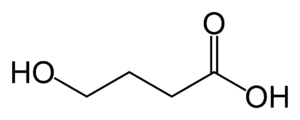 Image via Wikipedia Okay I'm not going to write an article about woulda coulda shoulda. But Jazz was shot down today heavily by the FDA by a vote of 20-No 2 Yes. Mostly due to concerns over abuse of the JZP-6 sodium oxybate drug which is highly similar to GHB. GHB was banned altogether by the FDA in 1990 and is viewed to have no medical benefits in the US.
Image via Wikipedia Okay I'm not going to write an article about woulda coulda shoulda. But Jazz was shot down today heavily by the FDA by a vote of 20-No 2 Yes. Mostly due to concerns over abuse of the JZP-6 sodium oxybate drug which is highly similar to GHB. GHB was banned altogether by the FDA in 1990 and is viewed to have no medical benefits in the US.
Labels:
Biotechnology and Pharmaceuticals,
FDA,
Gamma-Hydroxybutyric acid,
Jazz Pharmaceuticals,
Top Performing BioPharma Stocks,
Xyrem
8/18/10
Top Performing Biotech and Pharmaceuticals for Wednesday August 18th
 Image via CrunchBase Today was a good day for recommendations and Friday gets even better with Jazz Pharmaceuticals presenting August 20th to the FDA for their Fibramyalgia medication JZP-6 sodium oxybate. Jazz had huge gains this morning as the FDA handed out their review of the clinical trial. Jazz shot up around the 11.90 mark and has been so oversold that right now it's pushing 10.49 in afterhours trading. It has been All Over The Map Today. Speaking of all over the map, BSD Medical exploded today with their medical device approval from the FDA. Just exploded all over the map. What a day.
Image via CrunchBase Today was a good day for recommendations and Friday gets even better with Jazz Pharmaceuticals presenting August 20th to the FDA for their Fibramyalgia medication JZP-6 sodium oxybate. Jazz had huge gains this morning as the FDA handed out their review of the clinical trial. Jazz shot up around the 11.90 mark and has been so oversold that right now it's pushing 10.49 in afterhours trading. It has been All Over The Map Today. Speaking of all over the map, BSD Medical exploded today with their medical device approval from the FDA. Just exploded all over the map. What a day. Jazz was disappointing in the fact that everyone sold. Everyone is scared to death to leave it in the market for the FDA decision. Shoot the dice and let it roll sometimes. But we know where most people stand on this one. I'm taking my profits and waiting for tomorrow where I kick myself for selling. This one's a no-brainer in my department for Approval tomorrow.
Top Performing Biotech Stocks for Wednesday August 18th
Labels:
Biotechnology and Pharmaceuticals,
Endo Pharmaceuticals,
FDA,
Jazz Pharmaceuticals,
Top Performing Biotech Pharmaceuticals Stocks
BSD Medical receives FDA clearance for MTX-180
 Image via CrunchBase SALT LAKE CITY, Aug 18, 2010 (BUSINESS WIRE) -- BSD Medical Corporation /quotes/comstock/15*!bsdm/quotes/nls/bsdm (BSDM 1.82, +0.69, +61.06%) (Company or BSD) today announced that the U.S. Food and Drug Administration (FDA) has granted the Company a 510(k) clearance to market its MicroThermX Microwave Ablation System (MTX-180) for ablation of soft tissue. Clearance from the FDA of BSD's 510(k) Premarket Notification submission authorizes the commercial sale of the MTX-180 in the United States. The MTX-180 was designed to provide a higher power, optimized system targeted to the growing therapeutic interventional and surgical oncology market.
Image via CrunchBase SALT LAKE CITY, Aug 18, 2010 (BUSINESS WIRE) -- BSD Medical Corporation /quotes/comstock/15*!bsdm/quotes/nls/bsdm (BSDM 1.82, +0.69, +61.06%) (Company or BSD) today announced that the U.S. Food and Drug Administration (FDA) has granted the Company a 510(k) clearance to market its MicroThermX Microwave Ablation System (MTX-180) for ablation of soft tissue. Clearance from the FDA of BSD's 510(k) Premarket Notification submission authorizes the commercial sale of the MTX-180 in the United States. The MTX-180 was designed to provide a higher power, optimized system targeted to the growing therapeutic interventional and surgical oncology market. 8/8/10
Nanoshell particles
 Image via Wikipedia Nanoparticle Cancer Treatments 100% Eradication
Image via Wikipedia Nanoparticle Cancer Treatments 100% Eradicationby James Rickman
Nanoshell therapy is comprised of three components:
- an off-the-shelf near infrared laser source
- an off-the-shelf interstitial fiber optic probe delivers the laser energy to a site near or inside the tumor
- the investigational nanoshell particles, a near-infrared absorbing, inert material designed to absorb and convert the laser energy into heat.
Labels:
Biotechnology,
cancer,
cancer therapy,
cancer treatment,
clinical,
FDA,
infrared,
Management of cancer,
medicine,
nanoparticle,
nanoshell,
nanotechnology,
trials
9/15/09
Salix Pharmaceutical Shares Soar 50%
Salix Pharmaceuticals today saw a 50% share increase based on Phase III clinical trial results. Salix (Nasdaq: SLXP) already sells rifaximin as a treatment for travelers’ diarrhea. That product, marketed with the name Xifaxan, provided $26.3 million of Salix’s $52.2 million in second-quarter revenue. The company is seeking FDA approval to market the drug for a different indication. On a side note, Salix Pharmaceuticals will market METOZOLV ODT under a licensing agreement with Wilmington Pharmaceuticals from recent approval on Sept 9, 2009.
Bill Forbes, Salix’ senior vice president and chief development officer, said in a statement that if approved, rifaximin would be the first new option on the market for managing HE in more than 30 years. Salix estimates that HE represents a $600 million market in the United States.
Rifaximin also has been granted an orphan drug designation by the FDA for treatment of HE. That designation provides seven years of marketing exclusivity in the United States if the drug gains FDA approval.
Salix Pharmaceuticals, Ltd. (NASDAQ: SLXP) announced on August 25, 2009, the FDA has accepted for filing and designated for Priority Review the Company's New Drug Application for rifaximin tablets 550 mg for the maintenance of remission of hepatic encephalopathy. Additionally, the FDA has informed the Company of its plan to schedule an Advisory Committee meeting in late February 2010 to discuss the application.
A Priority Review classification is granted to drugs offering major advances in treatment, or providing a treatment where no adequate therapy exists. Based on this classification, the FDA has issued an action date of December 24, 2009 under the Prescription Drug User Fee Act. However, the convening of an Advisory Committee in late February 2010 signals the December 24, 2009 action date will be delayed.
Notably Pepcid was acquired by Merck in February 2007
"We are extremely pleased with the outcome of our pivotal Phase 3 trials of rifaximin in the treatment of non-constipation irritable bowel syndrome," stated Bill Forbes, Pharm.D., Senior Vice President and Chief Development Officer, Salix Pharmaceuticals. "Irritable bowel syndrome, characterized by abdominal pain, bloating and altered bowel habits, is one of the most common chronic medical conditions. Non-constipation IBS comprises the most common forms of IBS by including patients that have either diarrhea-predominant or diarrhea-constipation alternating symptoms. Based on the most current understanding of IBS, TARGET 1 and TARGET 2 were designed to investigate the utility of rifaximin, a broad spectrum, minimally absorbed, gut-selective antibiotic, with minimal side effects, in relieving the symptoms of IBS by altering the bacteria believed to be responsible for creating the symptoms. TARGET 1 and TARGET 2 will serve as the confirmatory trials for the Company's New Drug Application seeking marketing approval for rifaximin as a treatment option in this condition which is associated with widespread prevalence, incapacitating symptoms and substantial medical costs. The Company is targeting to submit the NDA during the first half of 2010."
Total Assets were 377M. Total Liabilities were 137M. Total Liabilities and stock holder's equities were 377M with 63M in long term debt. For 6 months Revenue was 97M with a net loss of 29M. Net income from 2008 annual data was a loss of 47M on 178M in total evenue. Looking ahead to the third quarter of 2009, Salix expects to recognize a loss of approximately $0.30 per share and product revenue of about $60 million. Wall Street analysts expect the company to lose $0.28 per share on revenue of $60.43 million.
So financially they have been struggling a bit but most clinical trials are very expensive. With this approval and the NDA filing in Feb 2010 maybe they can turn the corner and make a substantial profit. I see the stock dropping a bit from the current volatility and hype but it should prove to be a profitable trade in the future. Most of their clinical trials are out of the way and they are an aggressive company acquiring the rights for future Gastrointestinal products.
Related articles
Bill Forbes, Salix’ senior vice president and chief development officer, said in a statement that if approved, rifaximin would be the first new option on the market for managing HE in more than 30 years. Salix estimates that HE represents a $600 million market in the United States.
Rifaximin also has been granted an orphan drug designation by the FDA for treatment of HE. That designation provides seven years of marketing exclusivity in the United States if the drug gains FDA approval.
Salix Pharmaceuticals, Ltd. (NASDAQ: SLXP) announced on August 25, 2009, the FDA has accepted for filing and designated for Priority Review the Company's New Drug Application for rifaximin tablets 550 mg for the maintenance of remission of hepatic encephalopathy. Additionally, the FDA has informed the Company of its plan to schedule an Advisory Committee meeting in late February 2010 to discuss the application.
A Priority Review classification is granted to drugs offering major advances in treatment, or providing a treatment where no adequate therapy exists. Based on this classification, the FDA has issued an action date of December 24, 2009 under the Prescription Drug User Fee Act. However, the convening of an Advisory Committee in late February 2010 signals the December 24, 2009 action date will be delayed.
"We are extremely pleased with the outcome of our pivotal Phase 3 trials of rifaximin in the treatment of non-constipation irritable bowel syndrome," stated Bill Forbes, Pharm.D., Senior Vice President and Chief Development Officer, Salix Pharmaceuticals. "Irritable bowel syndrome, characterized by abdominal pain, bloating and altered bowel habits, is one of the most common chronic medical conditions. Non-constipation IBS comprises the most common forms of IBS by including patients that have either diarrhea-predominant or diarrhea-constipation alternating symptoms. Based on the most current understanding of IBS, TARGET 1 and TARGET 2 were designed to investigate the utility of rifaximin, a broad spectrum, minimally absorbed, gut-selective antibiotic, with minimal side effects, in relieving the symptoms of IBS by altering the bacteria believed to be responsible for creating the symptoms. TARGET 1 and TARGET 2 will serve as the confirmatory trials for the Company's New Drug Application seeking marketing approval for rifaximin as a treatment option in this condition which is associated with widespread prevalence, incapacitating symptoms and substantial medical costs. The Company is targeting to submit the NDA during the first half of 2010."
Looking over Salix's Financials can be found here: SALIX PHARMACEUTICALS LTD Form 10-Q August 10, 2009
Total Assets were 377M. Total Liabilities were 137M. Total Liabilities and stock holder's equities were 377M with 63M in long term debt. For 6 months Revenue was 97M with a net loss of 29M. Net income from 2008 annual data was a loss of 47M on 178M in total evenue. Looking ahead to the third quarter of 2009, Salix expects to recognize a loss of approximately $0.30 per share and product revenue of about $60 million. Wall Street analysts expect the company to lose $0.28 per share on revenue of $60.43 million.
So financially they have been struggling a bit but most clinical trials are very expensive. With this approval and the NDA filing in Feb 2010 maybe they can turn the corner and make a substantial profit. I see the stock dropping a bit from the current volatility and hype but it should prove to be a profitable trade in the future. Most of their clinical trials are out of the way and they are an aggressive company acquiring the rights for future Gastrointestinal products.
Related articles
- Salix Pharmaceuticals Announces Extension of XIFAXAN550 sNDA PDUFA Goal Date to March 7, 2011 (eon.businesswire.com)
- Salix Announces FDA Acceptance for Filing and Priority Review Designation For XIFAXAN550 sNDA For the Treatment of Non-Constipation Irritable Bowel Syndrome (eon.businesswire.com)
- Salix Pharmaceuticals Announces NDA Submission For XIFAXAN550 For Treatment of Non-Constipation Irritable Bowel Syndrome (eon.businesswire.com)
Related articles
Labels:
FDA,
IBS,
Irritable bowel syndrome,
New Drug Application,
Phase 3,
rafaximin,
Salix Pharmaceuticals,
SLXP
9/4/09
Glaxo's cancer vaccine inches toward approval--Merck's Cervarix vs GSK Gardasil.

By MATTHEW PERRONE - AP Business Writer
Published: Fri, Sep. 04, 2009
WASHINGTON -- Federal regulators said Friday that a GlaxoSmithKline vaccine prevents the leading cause of cervical cancer in women, bringing the company one step closer to competing with Merck's blockbuster Gardasil, which has controlled the U.S. market for three years. GlaxoSmithKline(GSK) Google Finance
In documents posted online, the Food and Drug Administration said Cervarix - Glaxo's vaccine against human papilloma virus or HPV - successfully blocked the two leading strains of the virus nearly 93 percent of the time.
But even as the British drugmaker moves closer to competing in the U.S., Merck is poised to begin marketing Gardasil to boys and men. In a separate review, the FDA said that vaccine prevented genital warts in males 90 percent of the time.
The agency will ask a panel of vaccine experts next week to weigh in on both vaccines. The FDA is not required to follow the group's advice, though it usually does.
While Merck has suggested the approval for boys could double the potential market for Gardasil, Leerink Swann analyst Seamus Fernandez said the benefit likely will be minimal.
Genital warts caused by HPV usually clear up by themselves, and the cancers caused by the virus are extremely rare in men.
"If it's not preventing something serious like cervical cancer and there are questions about safety, I think parents' acceptance of the vaccine in young boys might be less urgent than for their girls," said Fernandez.
Glaxo has won a number of government contracts for Cervarix in Europe, but its U.S. launch was delayed in 2007 when the FDA said it needed more data about the vaccine. Earlier studies of Cervarix showed a higher number of muscular and neurological problems among patients who used the vaccine compared with an alternate treatment.
The FDA said Friday that outside experts have now determined Cervarix did not cause those problems.
"The conclusion in the case of each of these efforts was that the data are not sufficient to establish a link," the agency said in its review.
FDA reviewers also noted a slightly higher rate of miscarriages in women taking Cervarix, but said "the data do not establish a causal relationship."
Glaxo wants its vaccine approved for girls and women ages 10 to 25. The FDA is expected to make a decision on the vaccine later this year, but analysts say London-based Glaxo may still have trouble making inroads into the U.S. market.
Gardasil and Cervarix both defend against HPV strains 16 and 18, which cause about 70 percent of cervical cancer cases. But Merck's vaccine also defends against two other HPV types that cause 90 percent of genital warts, something Cervarix does not target.
The most common side effects with both vaccines were pain and swelling at the injection site.
Gardasil became an early success story for Merck after its 2006 launch, achieving sales that are rare for a vaccine. The company has sold about 50 million doses worldwide, with more than $1.4 billion in revenue last year.
But sales have been slowing amid questions about the longevity of the vaccine's effect and its price tag of nearly $400. In the most recent quarter, Gardasil sales fell to $268 million, down 18 percent from the prior year period.
A government-funded study last year found that the HPV vaccine is cost-effective for young teens, but not for women in their 20s. And labeling for the vaccine notes that the "duration of protection of Gardasil has not been established." Company trials have only tracked immunity out to five years after receiving the three-injection regimen.
Whitehouse, N.J.-based Merck will make the case on Sept. 9 for approving the vaccine for boys and men ages 9 to 26.
Labels:
Cervarix,
Cervical cancer,
FDA,
Gardasil,
GlaxoSmithKline,
HPV vaccine,
Human papillomavirus,
Merck
8/27/09
Mylan Pharmaceuticals Stock Analysis


Mylan Pharmaceuticals (Nasdaq:MYL) keeps coming up on my radar so I would like to take the time to analyze the stock and give some recommendations on it. Mylan is approaching its 52 week high so is it a buy or not? In October 2008, they reached a 52 week low of 5.75 and today is trading at 14.88. So that old adage of Buy Low-Sell High is coming into play. In these economic times many rules of trading are finding exceptions to that rule. Mylan's Financial picture can be found on Google Finance here. I see a good exception to Mylan's future, and see the company's stock rising based on FDA approvals that have come in over the past few months.
I really like the generic market right now. With patents expiring you can literally gauge how much a generic will make based on the patents expiring in the next few months. These generics are the hyenas in a pack of lions that have a good way of scavenging for their food in the Serengeti. The hyena has a distinct relationship with the Lions. The lions kill much of the food the hyena scavenges. But then there are vultures too so we will end this charade and get to the point. They may not come up with the drugs but they are definitely going to make a nice profit from them. But the competition is fierce these days. Some competitors in the generic market include Watson (WPI), Teva (TEVA) , Dr Reddy's Labs (RDY), Hi-Tech Pharmacal (HITK), Par Pharmaceuticals (PRX), and Caraco (CPD).
Recent news has cleared Mylan's name from their Morgantown, WV 483 letter fiasco. Mylan's stock went from 13.85 to 12.10 based on the news on July 27th. The Post Gazette article can be found here titled Adverse reaction: The FDA probes quality control at Mylan. It is funny how quick we come to judgement over initial accusations. With that said, the Pittsburgh Post Gazette published allegations of profound quality control issues that got the attention of the FDA to investigate their plant. No 483 letter was issued and Mylan has since sued the Newspaper over the SOP deviation document stories. FDA cleared Mylan and Mylan Pharmaceuticals Receives Final FDA Report and Reaffirms Its 48-Year Exemplary Record of Quality on August 13th.
But to run a business in a lower economic area like West Virginia can be profitable, if they can handle their quality control issues. They can pay employees less based on the economic value of the area and can make more money. I have seen this first-hand by working outside of Raleigh in a rural area and finding out how much less we were making than those in the RTP area. Anyways, they choose these sites for a reason. I love West Virginia and have rafted the Upper and Lower Gauley myself. People in West Virginia definitely need the jobs.
Anyways, is this company going to make money or not? I say yes. Going through PR Newswire recently, Mylan continues gaining FDA approval for marketing for key generics. Here is a list compiled from the last few months for FDA approvals.
1. August 27, 2009 Mylan Begins Marketing First Generic Version of BenzaClin(R) Acne Treatment
2. August 18, 2009 Mylan's Matrix Receives First and Only Tentative FDA Approval Under PEPFAR for Generic Version of Atripla(R) HIV Treatment
3. August 10, 2009 Mylan Receives FDA Approval for Generic Version of Migraine Treatment Imitrex(R)
4. July 20, 2009 Mylan Receives FDA Approval for Additional Strengths of the Antipsychotic Haloperidol---> Key News from the article. Currently, Mylan has 119 ANDAs pending FDA approval representing $84.7 billion in annual brand sales, according to IMS Health. Thirty-five of these pending ANDAs are potential first-to-file opportunities, representing $16.6 billion in annual brand sales, according to IMS Health.
5. July 16, 2009 Mylan Receives FDA Approval for Generic Version of Thyroid Deficiency Treatment Cytomel
6. July 7, 2009 Mylan Receives FDA Approval for Generic Version of Prostate Cancer Treatment Casodex
7. June 17, 2009 Mylan Receives FDA Approval for Additional Strength of Generic Restoril(R)
8. May 29, 2009 Mylan Receives Tentative FDA Approval for Generic Version of Singulair(R)
9. May 7, 2009 Mylan Receives Final FDA Approvals for Generic Versions of Anti-Rejection Medication CellCept(R)
10. April 14, 2009 Mylan Confirms First-to-File Patent Challenge Relating to Xeloda(R) Cancer Treatment
But with the good I would like to investigate their financials a little closer. They have a Market Cap of 4.54 Billion. For the six months ended June 30, 2009, Mylan reported total revenues of $2.48 billion compared to $2.28 billion in the same prior year period. This represents an increase of $199.3 million or 8.8%. Net revenues increased $174.5 million, while other revenues increased $24.8 million. The increase in net revenues is due to higher third-party sales in all three of the Company's segments. The Generics Segment accounted for the majority of the increase ($131.3 million) followed by the Matrix Segment ($25.2 million) and the Specialty Segment ($18.0 million). On a constant currency basis, total revenues increased by approximately 17%. Their August 3rd quarterly report can be found here.
Revenue is up and gross profit is up too. However, so was their operating expenses. Earnings per share were 0.19, slightly down from Q1, but not a loss. That is good news. I see their generic market for HIV medications as their key growth. President Clinton and Mylan Chairman and CEO Robert J. Coury Announce New Agreement to Lower Price of Treatment for Patients with Drug-Resistant HIV in Developing Countries.
Labels:
483 Letter,
FDA,
Generics,
HIV,
Morgantown,
MYL,
Mylan Pharmaceutical,
Pittsburgh Post-Gazette,
President Clinton,
WV
8/2/09
Savient Pharma Receives Bad News Concerning KRYSTEXXA


This was on PR Newswire for Savient Pharma. Tough news for their Gout Treatment. Expect a huge downfall in stock price this week. In addition to Savient, there are other competitors out there with approvals so this is really bad news for Savient. However, I say wait for it to drop and buy it up on its low point. This lowpoint will probably will be reached sometime this week.
Uloric, from Takeda Pharmaceutical, was approved in February. Expect a huge swing in Takeda Pharma this week with this unexpected news. Previously, an advisory committee approved the drug, Krystexxa, so this is a shock for Savient as it was advised on a 14-1 vote. This just goes to show how volatile and risky Biotech stocks can be. Now this can be turned around, but it will take some tweaking in the lab and further involvement with Contract Manufacturing of the medication. Again tough news for Savient Pharma.
Also, another treatment for Gout was approved just last week. The company is Mutual Pharmaceutical out of Philadelphia, PA. FDA approved Colchicine for Acute Gout on July 30,2009. Double Whammy time for Savient. But I expect them to bounce back but we will have to see.
I'm including the PR newswire article at the end of this post.
Savient Pharmaceuticals Receives Complete Response Letter from U.S. Food and Drug Administration for KRYSTEXXA(TM)
Savient to Host Conference Call on Monday, August 3, 2009 at 8:00am
EAST BRUNSWICK, N.J., Aug. 2 /PRNewswire-FirstCall/ -- Savient Pharmaceuticals, Inc. (Nasdaq: SVNT) today announced that the Company has received a complete response letter from the U.S. Food and Drug Administration (FDA) stating that the FDA can not at this time approve the Company's Biologics License Application (BLA) for KRYSTEXXA(TM) (pegloticase) as a treatment for chronic gout in patients refractory to conventional therapy.
The complete response letter from the FDA cites deficiencies with the chemistry, manufacturing and controls (CMC) section of the BLA and also provided the current draft of the proposed labeling and further guidance regarding a Risk Evaluation and Mitigation Strategy (REMS) (Medication Guide and Communication Plan). The Company intends to immediately request a meeting with the FDA to discuss and clarify the issues raised in the complete response letter. Under FDA regulations, the Company believes that this meeting is deemed a "Type A" meeting, meaning that the FDA would meet with the Company within 30 days of its receipt of the meeting request.
One of the issues raised by the FDA in the complete response letter addresses a change made by the Company in the proposed process for manufacturing KRYSTEXXA for commercial use. The FDA has concluded that the comparability data submitted for the material manufactured using the proposed commercial manufacturing process was not adequate to demonstrate that it was representative of the material used to establish the safety and efficacy of KRYSTEXXA in its Phase 3 clinical trials. The FDA stated that the Company has the option of either reverting to and validating the manufacturing process used to produce KRYSTEXXA for the Phase 3 clinical trials or conducting additional comparability clinical trials to support the use of KRYSTEXXA manufactured using the proposed commercial manufacturing process. The Company currently expects that it will seek to address this issue by reverting to and revalidating the manufacturing process used to produce KRYSTEXXA for the Phase 3 clinical trials.
The complete response letter also stated that the FDA has determined that a REMS is necessary for KRYSTEXXA consisting of:
A Medication Guide to ensure the safe and effective use of KRYSTEXXA by patients,
A Communication Plan directed to healthcare providers likely to prescribe KRYSTEXXA to support the dissemination of information about the risks of severe infusion reactions and possible anaphylaxis, the risk of severe adverse reactions in administering KRYSTEXXA to patients with glucose-6-phopshate dehydrogenase (G6PD) deficiency and major cardiovascular events, and
An Assessment Plan to monitor and assess the effectiveness of the Medication Guide and Communication Plan in communicating to patients and physicians an understanding of the risks of KRYSTEXXA treatment.
The complete response letter included additional CMC comments focused on tightening manufacturing parameters and narrowing analytical specifications associated with commercial production. The Company was also informed that its resubmission to the FDA in response to the complete response letter must include an update of safety data from all on-going studies. Additionally, the Company's drug substance manufacturer BTG-Israel has already provided a work plan to remediate observations arising from the FDA pre-approval inspection of BTG-Israel's manufacturing facility and a satisfactory inspection report is required prior to the approval of KRYSTEXXA.
"While our timeline for resubmission to the FDA is subject to a number of uncertainties, we currently believe that we can target completion of our resubmission for early 2010. We hope to have more clarity on the expected timeline after we meet with the FDA to discuss the complete response letter," stated Paul Hamelin, President of Savient Pharmaceuticals. "While we believe we have made substantial progress toward the potential final approval of KRYSTEXXA, we also have more work to do with the FDA to resolve these open issues. We are committed to work diligently to address these issues with a goal of obtaining final approval for KRYSTEXXA so we can provide this therapy to those chronic gout patients who are suffering from this crippling, debilitating disease and have no other treatment options."
The Company believes that its resubmission will respond to all of the deficiencies cited in the compete response letter and would lead to a new Prescription Drug User Fee Act expected action date of either two or six months after the date of the Company's resubmission, depending on the FDA's classification of the resubmission.
ABOUT KRYSTEXXA(TM)
KRYSTEXXA(TM) (pegloticase) is a PEGylated uricase enzyme intended for the treatment of chronic gout in patients refractory to conventional therapy. Gout refractory to conventional therapy occurs in patients who have failed to normalize serum uric acid and whose signs and symptoms are inadequately controlled with xanthine oxidase inhibitors at the maximum medically appropriate dose or for whom these drugs are contraindicated.
ABOUT SAVIENT PHARMACEUTICALS, INC.
Savient Pharmaceuticals, Inc. is a specialty biopharmaceutical company focused on developing and marketing pharmaceutical products that target unmet medical needs in both niche and broader specialty markets. Savient has developed one product: KRYSTEXXA(TM) (pegloticase) which is a PEGylated uricase enzyme intended for the treatment of chronic gout in patients refractory to conventional therapy. Savient has exclusively licensed worldwide rights to the technology related to KRYSTEXXA, formerly referred to as Puricase(R), from Duke University and Mountain View Pharmaceuticals, Inc. Savient also manufactures and supplies Oxandrin(R) (oxandrolone tablets, USP) CIII in the U.S. Further information on Savient can be accessed by visiting: http://www.savient.com. Puricase is a registered trademark of Mountain View Pharmaceuticals, Inc.
The previous advisory panel recommendation was found in the New York Times on June 16, 2009. The link to that article can be found at the end of this post.
The approval for Colchicine is found on the FDA's website. I've included it here.
FDA Approves Colchicine for Acute Gout, Mediterranean Fever
Agency also provides new information to physicians regarding safe use of drug
The U.S. Food and Drug Administration has approved Colcrys to treat acute flairs in patients with gout, a recurrent and painful form of arthritis, and patients with familial Mediterranean fever (FMF), an inherited inflammatory disorder. The medication’s active ingredient is colchicine, a complex compound derived from the dried seeds of a plant known as the autumn crocus or meadow saffron (Colchicum autumnale).
Colchicine has been used by healthcare practitioners for many years to treat gout but had not been approved by the FDA. The FDA has an initiative underway to bring unapproved, marketed products like colchicine under its regulatory framework. This initiative promotes the goal of assuring that all marketed drugs meet modern standards for safety, effectiveness, quality and labeling.
Physicians historically have given colchicine hourly for acute gout flares until the flare subsided or they had to stop treatment because the patient began experiencing gastrointestinal problems. A dosing study required as part of FDA approval demonstrated that one dose initially and a single additional dose after one hour was just as effective as continued hourly dosing for acute gout flares, but much less toxic. As a result, the drug is being approved for acute gout flares with the lower recommended dosing regimen.
The FDA is alerting healthcare professionals to this new dosing regimen and also warning about the potential for severe drug interactions when patients take colchicine.
The medicinal value of using colchicum was first identified in the first century A.D. and its use for treating acute gout dates back to 1810. Physicians have prescribed the medication since then. Although single-ingredient colchicine has not been approved by the FDA until now, a combination product containing colchicine and an agent that increased the excretion of uric acid in the urine was approved by the FDA in 1939.
FMF is the most common of the hereditary periodic fever syndromes and is characterized by recurrent episodes of fever, arthritis and painful inflammation of the lining layers of the lungs and abdomen. Though rare in the United States, it is more common in Mediterranean countries. Physicians have prescribed colchicine for FMF for many years based on studies showing that it reduced the frequency of attacks but use of colchicine for FMF had never been approved. With this approval, Colcrys becomes the first drug approved to treat FMF.
Colcrys is manufactured by Mutual Pharmaceutical Company, Inc., Philadelphia.
Labels:
Clinical trial,
FDA,
Gout,
Mutual Pharmaceutical Company,
Savient Pharmaceutical,
Takeda Pharmaceutical Company,
U.S. Food and Drug Administration,
Uric acid
Subscribe to:
Posts (Atom)




![Reblog this post [with Zemanta]](http://img.zemanta.com/reblog_e.png?x-id=7b965007-456e-4fcf-8f32-ad7e65762697)
![Reblog this post [with Zemanta]](http://img.zemanta.com/reblog_e.png?x-id=1b7f8182-d212-4b33-9583-1260ea1fbd8c)